The influenza virus that caused the 1918 pandemic mutated into variants, much like the novel coronavirus has done in the current pandemic, century-old virus samples reveal.
The discovery could help explain why later waves of the 1918 flu pandemic were worse than the first.
And while the results aren’t directly applicable to the COVID-19 pandemic, they do show that virus variants are to be expected — and that humanity can ultimately overcome them, one expert told.
“Those [viral infections] in the second wave look like they were better adapted to humans,” said study lead author Sébastien Calvignac-Spencer, an evolutionary biologist at the Robert Koch Institute in Berlin, Germany’s federal disease control and prevention agency. “Just like today, we wonder whether the new variations behaved differently or not than the original.”
To answer that question, Calvignac-Spencer and his colleagues found six human lungs that dated to the pandemic years of 1918 and 1919 and had been preserved in formalin in pathology archives in Germany and Austria.
The researchers determined that three of those lungs — two from young soldiers who had died in Berlin, and one from a young woman who had died in Munich — contained the 1918 influenza virus.
Pandemic flu
The virus responsible for the 1918 influenza pandemic still circulates today. But it was much deadlier then, mainly because humans today are descended from people who survived the infection more than a hundred years ago and so they’ve inherited some form of genetic immunity, Calvignac-Spencer said.
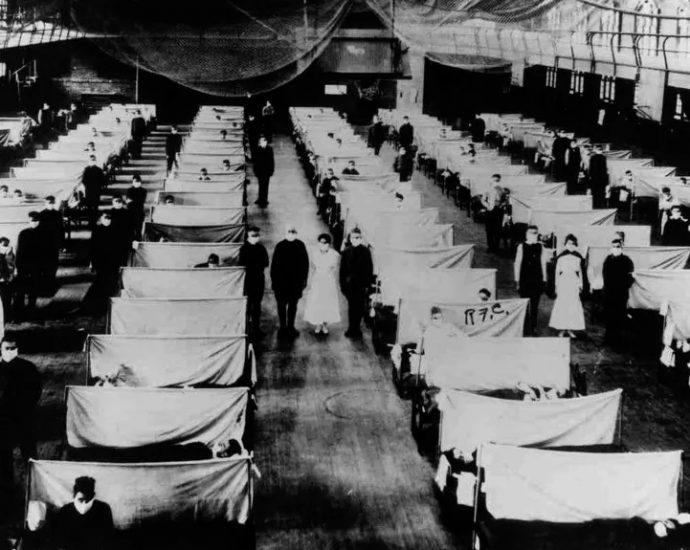
Estimates suggest this strain of influenza infected up to 1 billion people worldwide, when the global population was only 2 billion; between 50 million and 100 million people may have died in three successive waves, Calvignac-Spencer said.
The first wave of the pandemic, in early 1918, was less deadly than those that followed, and the preserved lungs of the two German soldiers who died in Berlin date from that time, he said.
The researchers extracted viral RNA from those samples to reconstruct about 60% and 90%, respectively, of the genomes of the flu virus that killed the soldiers.
The soldiers died on the same day, and the genomes of the virus that killed them showed almost no genetic differences between them, he said.
But the form of the flu virus found in their lungs had several genetic differences from the form of the virus that infected the young woman who died in Munich, presumably in a later wave of the pandemic. And they diverged even more from two genomes of the virus from Alaska and New York that dated from the second wave of the pandemic in late 1918, according to a new study published to the preprint database bioRxiv and which has not yet been peer-reviewed.
Deadly variants
The researchers compared the genomes from the U.S. and Germany and conducted lab studies with synthesized replicas of parts of the virus to learn how well different strains might have infected and replicated inside cells.
Their findings suggest that the virus mutated to become more effective between the first and later waves, by evolving to better overcome cellular defenses against infection, Calvignac-Spencer said.
The genetic mutations that popped up between the first and second waves may have made the virus better adapted to spreading among humans, rather than between birds, its natural hosts. Another mutation may have changed how the virus interacts with a human protein known as MxA, which helps orchestrate the body’s immune response to new pathogens.
Although scientists don’t know for sure how the genetic variations changed the behavior of the virus, “it’s predicted that these changes helped the virus to evade one of the mechanisms that [human] cells use to shut down influenza viruses,” he said.
The same process of viral evolution can be seen in the current COVID-19 pandemic, he said. “It’s interesting to make parallels — for example, the fact that there were multiple successive waves is a pattern which is intriguing.”
But scientists can learn more from the COVID-19 pandemic than they ever could about the 1918 flu pandemic, because science has now progressed so much, he said. “The more we can learn about the current pandemic, the more that can help us understand the past pandemic, rather than vice versa.”
A significant advance is that the researchers have been able to accurately sequence virus genomes in human tissue preserved in formalin for over 100 years — something that, until now, was thought to be very difficult.
Thanks to new techniques, “in the end, it was much easier than we expected,” he said; the same techniques mean it might now be possible to sequence virus genomes from human bodies infected with viruses buried in permafrost for up to 1,000 years, because the cold could help preserve DNA for much longer.
His team also wants to sequence virus genomes that might be preserved in the bodies of ancient Egyptian mummies, the earliest of which are around 5,000 years old. “They were prepared to stop biological processes, and that’s exactly what we want,” he said. “So we will give it a try as well.”